 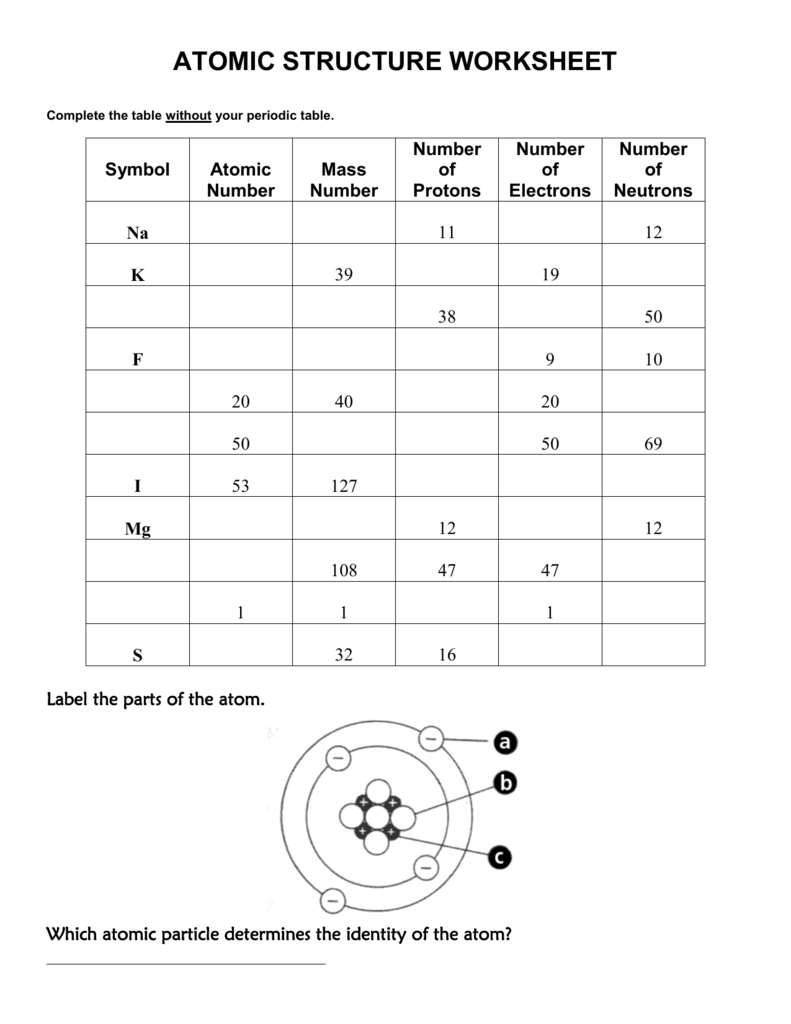
Basic Atomic Structure Worksheet H and the 1. The atomic model worksheet and key 3.Ītomic basics answer key part a. Draw five protons in the nucleus of the atom. Get thousands of teacher-crafted activities that sync up with the school year. You have all your materials.Ītomic basics worksheet answers along with valuable topics. 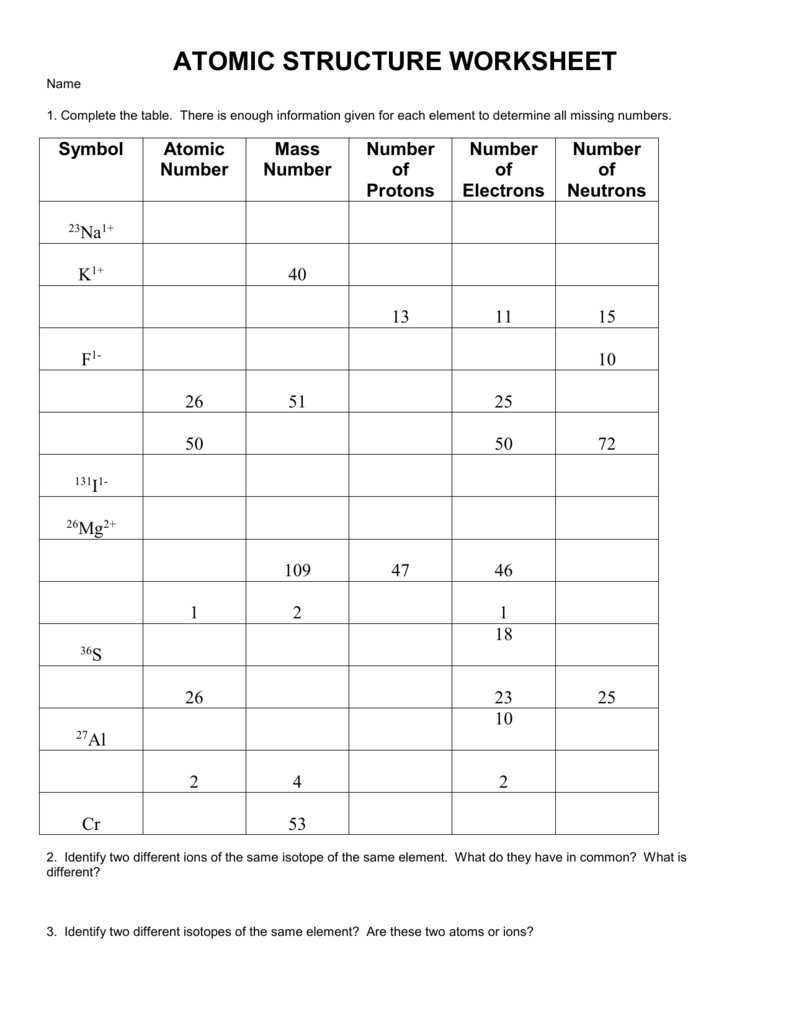
Draw six neutrons in the nucleus of the atom. Draw three electrons in the second energy level and label them with their charge.ĭraw two electrons in the first energy level and label them with their charge. Some of the worksheets for this concept are protons neutrons and electrons practice work answer key structure of matter work answers key ebook. Science worksheets by topic.īonding Basics Worksheet Answers Worksheets Are An Important Part Of Gaining Knowledge Of English In 2021 Ionic Bonding Covalent Bonding Worksheet Covalent Bonding Completed answer key can.Ītomic basics worksheet answer key part a. Chemical reactions largely involve atoms or groups of atoms and the interactions between their electrons.Label them with their charge. In general, electrons are easier to add or remove from an atom than a proton or neutron. The particles within an atom are bound together by powerful forces.Isotopes and ions of an atom with a constant number of protons are all variations of a single element. Varying the number of electrons results in ions. Varying the number of neutrons results in isotopes. The number of protons (also known as its atomic number) determines the element.Almost all of the mass of an atom is in its nucleus almost all of the volume of an atom is occupied by electrons.The average size of an atom is about 100 picometers or one ten-billionth of a meter. Electrons can also move between orbitals. 
Technically, an electron can be found anywhere within the atom, but spends most of its time in the region described by an orbital. Some electron shells resemble spheres, but others look more like dumb bells or other shapes. Simple models show electrons orbiting the nuclear in a near-circular orbit, like planets orbiting a star, but real behavior is much more complex. Electrons are organized into shells, which is a region where an electron is most likely found. 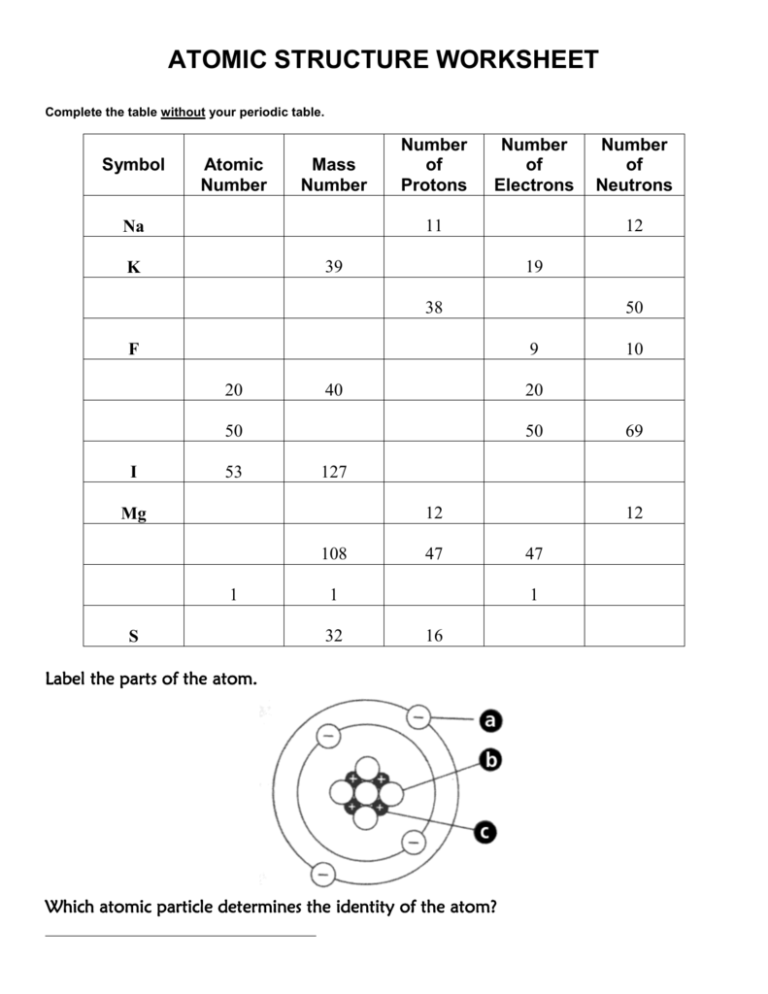
Electrons move around outside the nucleus.The nucleus carries a positive electrical charge. The nucleus of an atom contains protons and neutrons.The mass of a proton is 1840 times greater than the mass of an electron. The mass of a proton is essentially the same as that of a neutron. Protons and neutrons are about the same size as each other and are much larger than electrons.In other words, neutrons do not have a charge and are not electrically attracted to either electrons or protons. Electrons and protons are electrically attracted to each other. Like charges (protons and protons, electrons and electrons) repel each other. The charge of a proton and an electron are equal in magnitude, yet opposite in sign. Each proton has a positive electrical charge.Each electron has a negative electrical charge.Nuclear reactions, such as radioactive decay and fission, can break apart atoms. They do consist of parts, which include protons, neutrons, and electrons, but an atom is a basic chemical building block of matter. Atoms cannot be divided using chemicals. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
